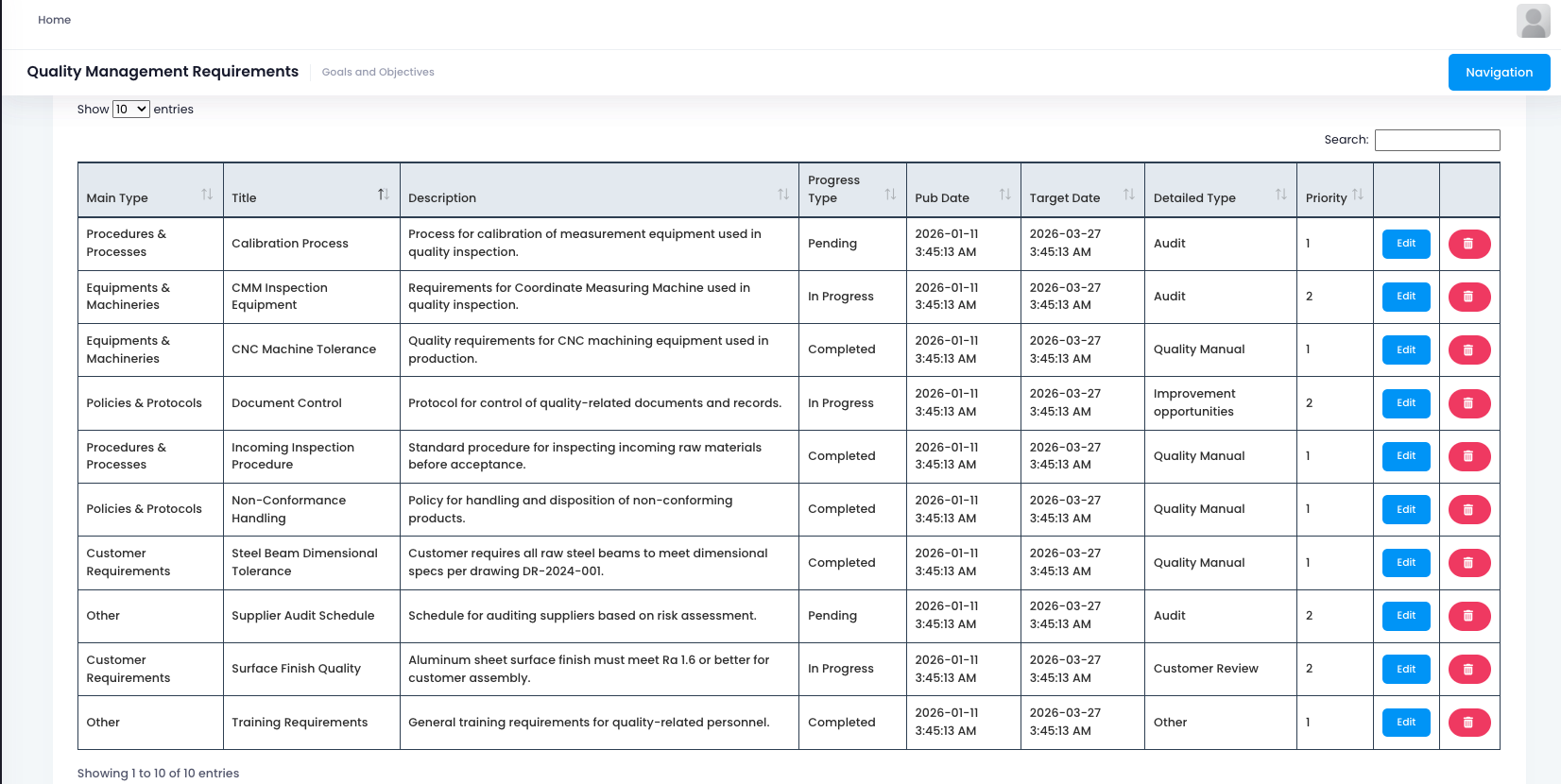
In today's competitive manufacturing landscape, quality isn't just a goal, it's a survival requirement. Today, we explore how a modern Quality Management System (QMS) can transform your quality control processes from reactive firefighting into proactive excellence that drives customer satisfaction and regulatory compliance.
Quality management is the foundation of any successful manufacturing operation. Without systematic control and visibility, businesses face constant risks of product defects, customer complaints, and regulatory non-compliance. Modern QMS solutions are now evolving beyond basic inspection tracking to provide intelligent, end to end quality management across the entire production lifecycle, from raw materials to finished goods delivery.
The cost of poor quality is staggering. Industry studies show that poor quality can cost manufacturers up to 15-20% of their total revenue. This includes direct costs like scrap, rework, and warranty claims, as well as indirect costs like lost customers, damaged reputation, and reduced market share. A robust QMS helps organizations identify and eliminate these costs systematically.
Unified Quality Dashboard: Real Time Visibility
The first step toward quality excellence is comprehensive visibility. A centralized Quality Dashboard provides a bird's eye view of all quality metrics, inspection results, and compliance status across all production lines, enabling data driven decision making and proactive quality management.
As shown in our dashboard, users can instantly see key quality indicators such as:
- Overall Quality Score: Aggregate quality performance metrics that provide immediate insight into production health
- Pass/Fail Rates: Real time tracking of inspection results across different product lines and stages
- Defect Counts: Current defect levels categorized by type, severity, and location
- Inspection Completion: Status of scheduled inspections and compliance checks
- Trend Analysis: Visual representations of quality trends over time to identify patterns
This high level visibility ensures that quality managers can quickly identify emerging trends, spot potential issues before they escalate, and make informed decisions about production adjustments, resource allocation, and process improvements. The dashboard also supports drill down capabilities, allowing users to investigate specific issues in detail.
Comprehensive Inspection Management: Standardized Processes
Effective quality control requires systematic, standardized inspection processes. Modern QMS provides detailed inspection management capabilities, allowing teams to plan, execute, and track inspections across all production stages with standardized procedures, digital documentation, and automated workflows.
This comprehensive inspection management enables teams to maintain consistent quality standards through:
- Standardized Inspection Plans: Predefined inspection procedures that ensure consistency across shifts, operators, and facilities
- Digital Checklists: Mobile enabled inspection checklists that guide operators through required steps and capture digital evidence
- Automated Scheduling: System generated inspection schedules based on production plans, risk assessments, and regulatory requirements
- Real Time Results Capture: Immediate recording of inspection results with photographic evidence and measurements
- Complete Audit Trails: Full documentation of who performed inspections, when, and with what results
By digitizing inspection processes, organizations eliminate paper based systems that are prone to errors, delays, and loss. Digital inspections ensure that all required checks are completed consistently, that results are immediately available for analysis, and that complete documentation is maintained for regulatory compliance and customer audits.
Key Benefits of Digital Inspections
- Reduce inspection time by up to 40% through mobile-enabled workflows
- Eliminate data entry errors with automatic result capture and synchronization
- Ensure 100% inspection coverage with automated scheduling and tracking
- Improve inspector productivity with standardized procedures and intuitive interfaces
- Maintain complete documentation for audits and compliance requirements
Defect Tracking and Root Cause Analysis: Continuous Improvement
When defects occur, rapid identification, analysis, and resolution are critical to preventing recurrence. Modern QMS provides sophisticated defect tracking and analysis capabilities, enabling root cause analysis, trend identification, and continuous improvement initiatives that drive long term quality excellence.
This defect tracking capability allows quality teams to systematically address quality issues through:
- Comprehensive Defect Classification: Categorization of defects by type, location, severity, and root cause for detailed analysis
- Pareto Analysis: Statistical identification of the most significant defect types to prioritize improvement efforts
- Root Cause Investigation: Structured tools and workflows for conducting thorough root cause analysis using methodologies like 5 Whys and Fishbone diagrams
- Corrective Action Tracking: Assignment and monitoring of corrective actions with deadlines and effectiveness verification
- Trend Monitoring: Continuous tracking of defect rates and patterns to measure improvement effectiveness
By maintaining detailed defect records and conducting systematic root cause analysis, organizations can move beyond simply detecting defects to preventing them entirely. This proactive approach reduces defect rates, improves first pass yield, and drives continuous quality improvement across all production processes.
Compliance and Certification Management: Regulatory Excellence
Regulatory compliance is a critical requirement for many industries, including automotive, aerospace, medical devices, and food production. Modern QMS provides comprehensive compliance management tools, ensuring that all quality processes meet industry standards and regulatory requirements while maintaining complete documentation for audits and certifications.
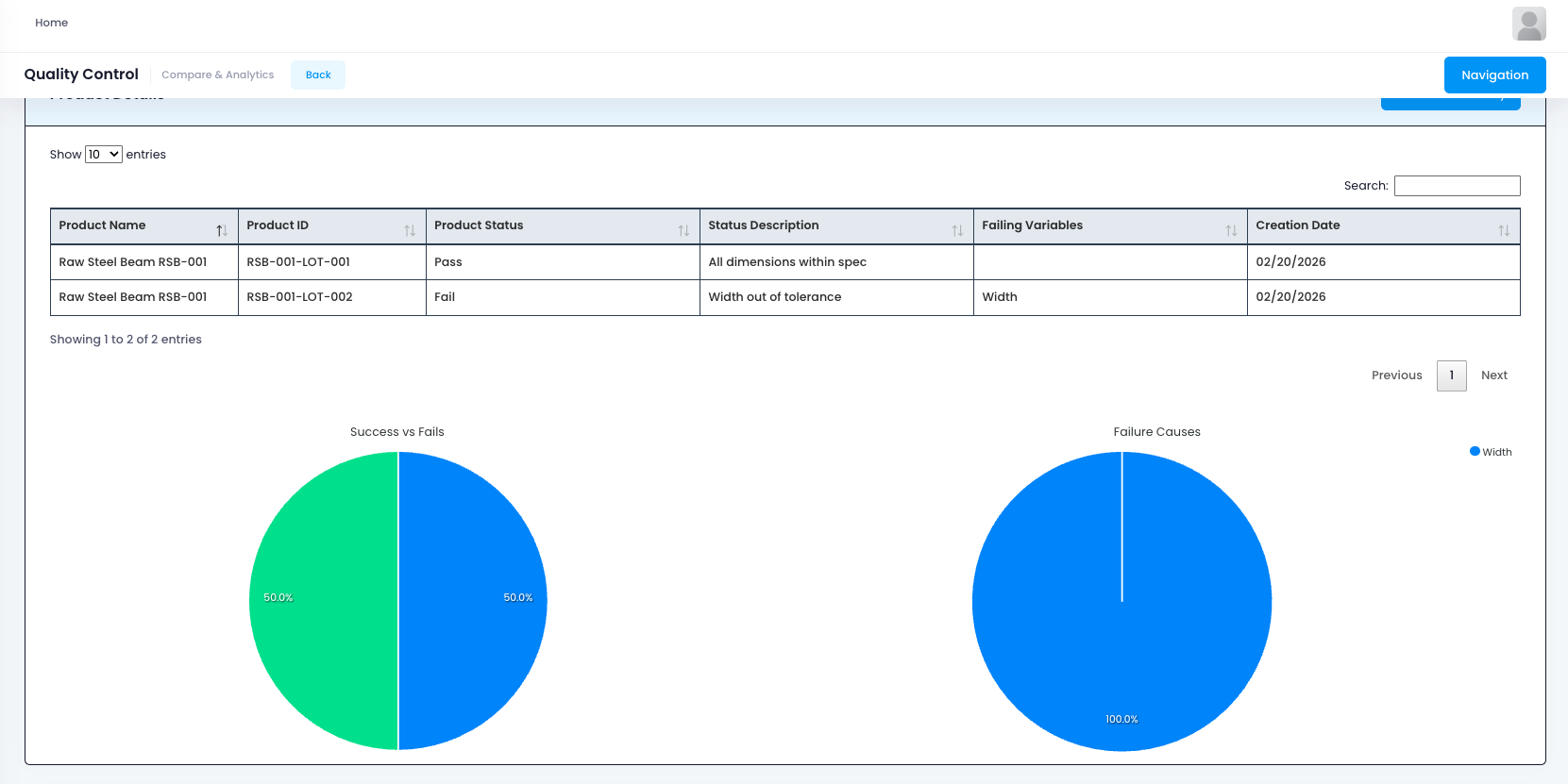
This compliance management capability ensures that quality processes align with industry standards such as:
- ISO 9001: Quality management system requirements for consistent product and service quality
- ISO 13485: Quality management system requirements for medical device manufacturers
- AS9100: Quality management system requirements for aerospace industry
- IATF 16949: Quality management system requirements for automotive industry
- FDA 21 CFR Part 820: Quality system regulations for medical devices
- Industry-Specific Standards: Customizable framework for any industry-specific requirements
The system provides automated alerts for upcoming audits, maintains complete documentation trails, and facilitates certification maintenance and renewal. This comprehensive compliance management reduces audit preparation time, ensures ongoing compliance, and provides confidence to customers and regulators that quality processes meet the highest standards.
Compliance Management Features
- Automated compliance gap analysis against industry standards
- Document control with versioning and approval workflows
- Training management to ensure personnel competency
- Internal audit scheduling and management
- Corrective and preventive action (CAPA) management
- Certification tracking and renewal reminders
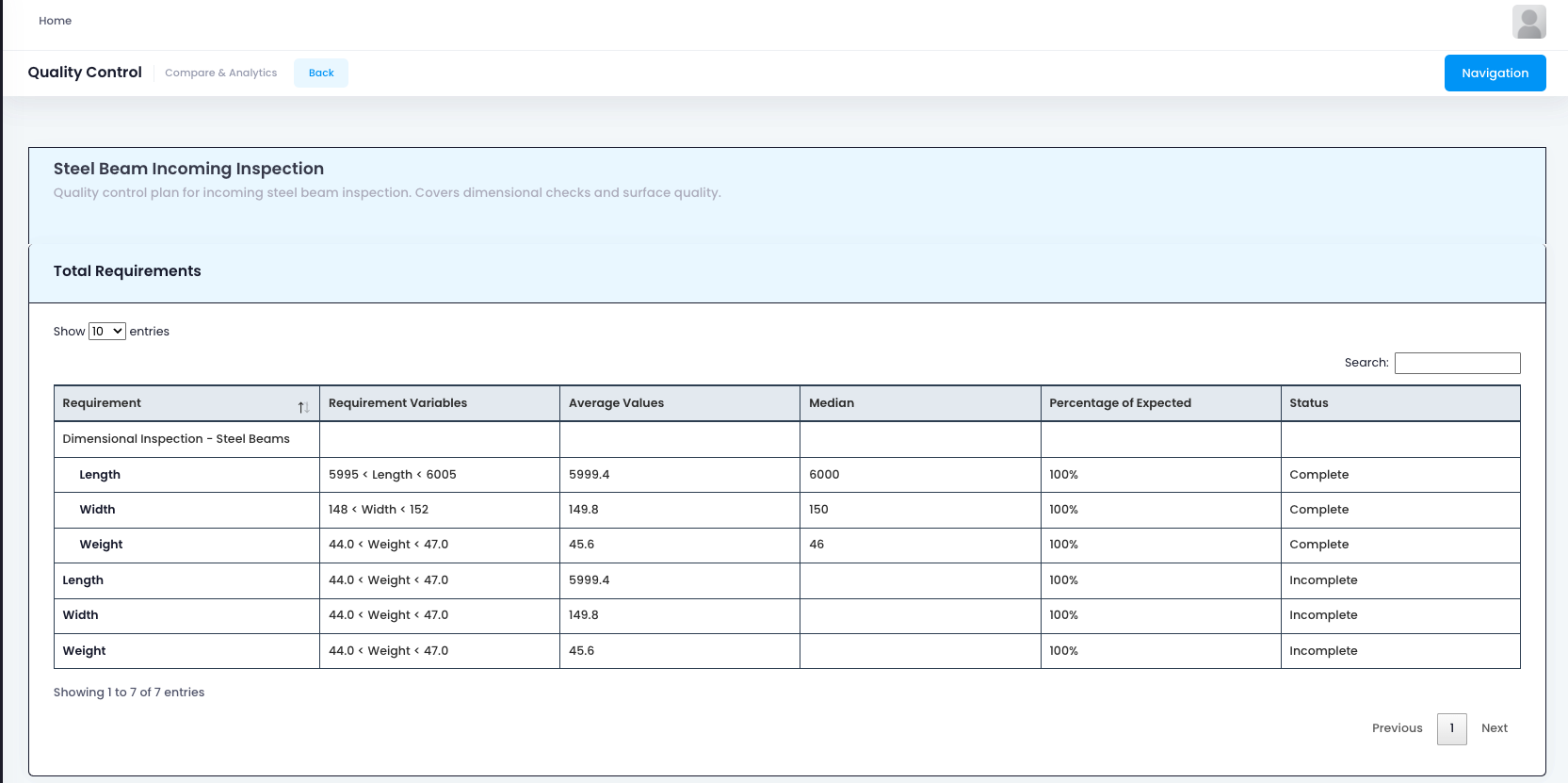
Advanced Quality Analytics: Predictive Insights
The next frontier in quality management is predictive analytics. Modern QMS solutions are increasingly incorporating AI driven insights to help businesses predict quality issues, optimize inspection schedules, and identify improvement opportunities before defects occur, transforming quality from reactive to proactive.
These advanced analytics capabilities enable businesses to leverage their quality data for strategic advantage through:
- Predictive Defect Modeling: Machine learning algorithms that predict defect likelihood based on process parameters and historical patterns
- Process Capability Analysis: Statistical analysis of process performance to identify capability gaps and improvement opportunities
- Optimized Inspection Scheduling: Data-driven recommendations for inspection frequency based on risk and performance
- Supplier Quality Analytics: Analysis of supplier performance to identify risks and improvement opportunities
- Cost of Quality Analysis: Comprehensive tracking of quality costs to identify improvement ROI
By leveraging these advanced analytics, organizations can move from reactive quality management, responding to defects after they occur, to proactive quality management that anticipates and prevents issues before they impact customers. This forward-looking approach helps reduce defect rates, improve first pass yield, and achieve consistent product quality while optimizing inspection resources and reducing overall quality costs.
Integration with Manufacturing Systems: End to End Quality
Modern QMS doesn't operate in isolation, it integrates seamlessly with other manufacturing systems to provide end to end quality visibility and control. This integration ensures that quality is considered at every stage of the product lifecycle, from design through production to delivery.
Key integrations include:
- Manufacturing Execution System (MES): Real time quality monitoring during production with automatic work order holds for quality issues
- Enterprise Resource Planning (ERP): Quality data integration with production planning, procurement, and financial systems
- Product Lifecycle Management (PLM): Quality requirements embedded in product design and engineering change processes
- Supply Chain Management: Supplier quality monitoring and incoming material quality tracking
- Customer Relationship Management (CRM): Quality complaint tracking and customer satisfaction monitoring
This integrated approach ensures that quality is not just a separate department but a fundamental consideration across all business processes. Quality data flows seamlessly between systems, enabling comprehensive quality management that considers the entire product lifecycle and all stakeholders.
Conclusion: Quality as a Competitive Advantage
In today's competitive marketplace, quality is not just a requirement, it's a competitive advantage. Companies that excel in quality management enjoy higher customer satisfaction, lower costs, stronger brand reputation, and greater market share. A modern Quality Management System provides the tools, processes, and insights needed to achieve quality excellence.
By adopting these modern Quality Management System practices, companies can:
- Reduce defect rates and improve first pass yield
- Lower quality costs through prevention rather than correction
- Ensure regulatory compliance and maintain certifications
- Build customer trust and loyalty through consistent quality
- Drive continuous improvement through data driven insights
- Achieve competitive advantage through quality excellence
The result is a quality-focused organization that delivers consistent, reliable products while continuously improving processes and reducing waste. Quality becomes not just a department but a culture that permeates the entire organization, driving excellence at every level.