At a Glance
Quality data collected in real-time, nonconforming products automatically blocked
Inspection processes digitized, paper records fully eliminated
Quality issue closed-loop management, end-to-end traceability from detection to corrective action
Supplier quality collaboration, incoming material issues controlled proactively
Customer Profile
A medical device manufacturer with annual revenue of approximately USD 300 million and 1,200 employees, primarily engaged in the research, development, and production of Class III implantable medical devices. The company’s products require multiple certifications including FDA, CE, and NMPA, demanding an exceptionally high quality system standard. The production process involves hundreds of quality control points, and inspection records had long relied on paper forms, requiring weeks of documentation preparation before each audit.
Quality Buried Under Paper
Inspectors filled out forms, signed them, and filed them away. When the quality manager wanted to know if a batch passed, they had to search through paper. When auditors needed to review nonconformance records from a specific month, they had to search through paper. When customer complaints required traceability, again the search through paper began.
Paper is evidence of quality, but it is also a cage for quality. Evidence is trapped inside. Information cannot escape.
This company was once exactly that. Until QMS liberated quality from the paper pile.

Quality Data Collected in Real Time
Previously, inspectors performed checks on the production line, recorded results with pen and paper, and entered data into computers at the end of the shift. Quality data was always yesterday's news.
Now, inspectors use handheld terminals. They inspect on-site and enter results immediately. Whether a product passes or fails, data enters the system instantly. The production line sees a failure and stops automatically. The quality manager opens the system and sees the quality status of every batch in real time.
Quality is no longer past tense. It is present tense.

Inspection Processes Digitized
Incoming inspection, in-process inspection, and finished goods inspection were previously three independent processes with three separate sets of forms. Inspection standards lived in filing cabinets. Inspection records lived on paper. Inspection results were communicated manually.
Now, all inspection processes are defined in the system. Which items to inspect for which materials, which parameters to check for which operations, which criteria to apply for which products all are structured. Inspectors follow system-guided instructions. Standards are not forgotten. Items are not missed. Results are automatically aggregated.
When processes are standardized, quality no longer depends on individuals.

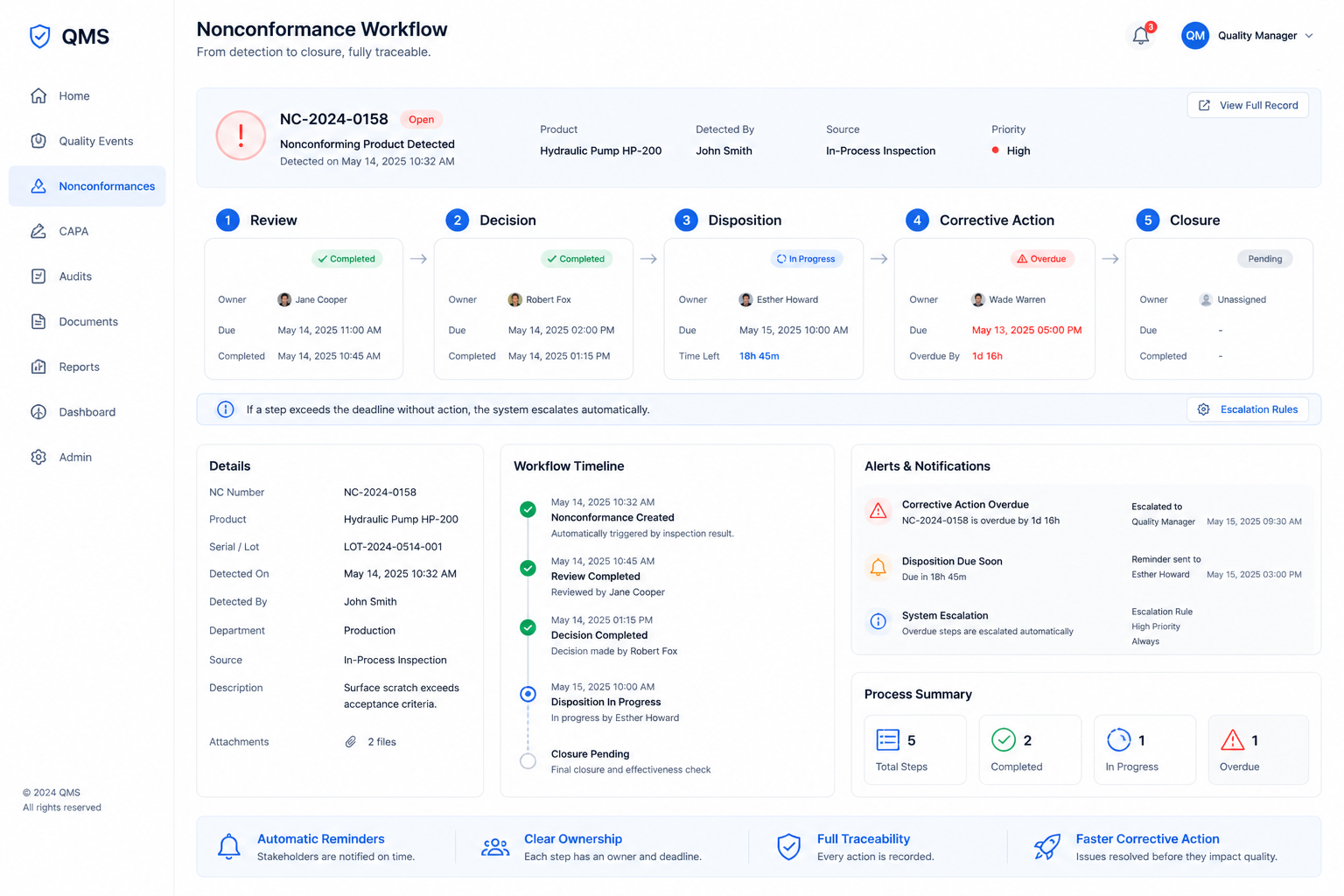
Nonconformance Closed Loop Management
When a nonconforming product was found, previously a red label was attached and the product was placed in a designated area, awaiting the quality manager’s handling. Disposition results were recorded in notebooks. Corrective actions were posted on bulletin boards.
Now, the moment a nonconforming product enters the system, a disposition workflow is automatically triggered. Who reviews, who determines, who disposes, who implements corrective actions each step has an assigned owner and deadline. If a step exceeds the deadline without action, the system escalates automatically.
From detection to closure, the entire nonconformance process is traceable. Corrective actions are no longer posted on walls. They are pushed to the responsible individuals.
Supplier Quality Collaboration
Previously, when incoming materials failed inspection, quality notified procurement, and procurement notified the supplier. Whether the supplier implemented corrective actions and how effective those actions were all depended on email exchanges back and forth.
Now, when incoming materials fail inspection, the system automatically notifies the supplier. Suppliers log into the system and see the nonconformance report, failure photos, and inspection data. Corrective action reports are submitted online.
Quality reviews them online. Only after approval can shipments resume.
Supplier quality is no longer one-way management. It is two-way collaboration.
Customer Testimonial
Before each FDA audit, we used to start preparing documentation two months in advance, sending dozens of cartons of paper to the meeting room. Now when auditors arrive, we open the system. All quality records are right there. Whatever they need, we search and pull it up on the spot. Audit pass rates are higher. Preparation time is shorter.